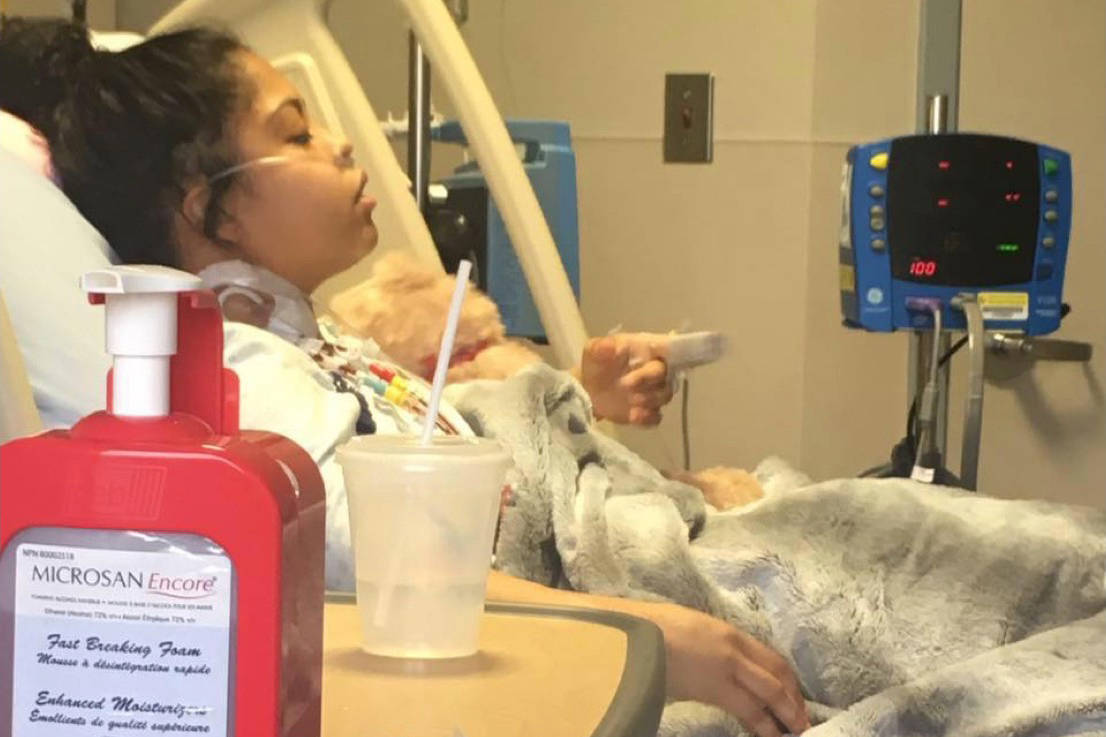
Shantee Anaquod is finally getting the life-saving drug she needs.
On Tuesday afternoon (Nov. 21), the province approved funding for the 23-year-old UBC student to receive Soliris, a drug that treats the rare disease Atypical Hemolytic Uremic Syndrome (aHUS) and costs $750,000 per year.
This comes after a Nov. 20 announcement by B.C. Health Minister Adrian Dix that the province will now cover the costs of Soliris on a case-by-case basis.
Shantee was diagnosed with aHUS shortly after Thanksgiving, and has been undergoing dialysis and plasma exchanges in hospital ever since. The ultra rare blood disease — which affects between 100 and 150 Canadians —causes a protein to be built that attacks the body’s own healthy cells, leading to blood clotting and kidney failure.
Previously, Shantee was denied coverage for the drug by both the B.C. government and her extended medical, despite other provinces like Ontario and Quebec covering the costs.
“After this was brought to his attention, the Minister asked the ministry to review coverage for Soliris provided in other jurisdictions, along with evidence from the past two years and advice from clinical experts in B.C.,” said Laura Heinze, manager of media relations and issues for the Ministry of Health.
“As a result, the expert expensive drugs for rare disease advisory committee co-ordinated by the Provincial Health Services Authority will now review recommended exceptional case-by-case requests for Soliris for aHUS and make funding recommendations to the Ministry. In making these decisions they will be using a framework proposed by BC’s leading nephrologists.”
READ MORE: Langley mom pleads for help in securing $750,000 life-saving drug for daughter
Shantee went through a review for Soliris coverage early Nov. 21, and by 1 p.m. was celebrating approval with her family.
“I was relieved, not only that there’s a chance of getting better, but also I didn’t have to fight anymore,” Shantee said. “It was exhausting.”
“It’s beyond amazing,” added Shantee’s mother, Jennifer, a Langley resident.
“There’s just no words.”
Shantee’s doctor was on standby will his cellphone all day waiting for a call from the committee.
“As soon as he knew, he came up — and I was going down to get some lunch for Shantee — and I saw him, he was on the phone, and I saw him give me a big thumbs up,” Jennifer said. “It’s just amazing.”
Shantee began her first treatment on Wednesday, and is hopeful she could be home from the hospital by Christmas.
“It was about 45 minutes and really easy to do and I woke up feeling better,” Shantee said. “I’m not sure if the medicine is kicking in yet or if it is a placebo effect from all the good news.”
Jennifer said the process to receive the Soliris has been frustrating, as Shantee’s condition worsened each day she did not have the drug.
She was scheduled to go for a kidney biopsy on Nov. 23 to determine if Soliris can help them regain function. If the damage is too severe, she will need a kidney transplant and will be required to take Soliris for that operation as well.
Also frustrating, the family received little feedback from the government, Jennifer added.
“Nobody’s received any response from the letters or emails,” she said.
“The only reason I spoke to anyone was that I forced myself into the press conference … Somebody tipped me off that it was happening, and I went to his (Dix) MLA office and forced myself in. That’s the only reason he’s had any contact with me, other than that I haven’t heard a word from anybody.
“And (Premier) John Horgan announced that they were working closely with the family. We haven’t heard from them at all. Like not once.”
Prior to her approval, Shantee’s family had a GoFundMe campaign to raise $100,000 for three doses of Soliris. Each dose costs $28,000, and they are administered every two weeks.
Now, the $37,475 that has been raised will go towards Shantee’s recovery and a kidney transplant, if needed.
“Because she’s been sick for so long, and so severely sick, it’s going to be a really long haul for her to get better,” Jennifer said.
COST OF SOLIRIS ‘EXCESSIVE’
On Tuesday afternoon, the ministry revealed the framework for patients seeking Soliris coverage. The patient must have a confirmed diagnosis of aHUS and evidence of active organ damage; they must have tried plasmapheresis as a treatment option first; they must be assessed throughout the treatment to ensure they are seeing improvement of their disease; and they must be assessed at six and 12 months to confirm if the drug therapy needs to continue.
“There are at least 50 patients across Canada who have received Soliris through exceptional public coverage today. We should be learning from those cases and leveraging the value of our public health care system to better inform provinces and territories struggling with these challenging decisions,” Heinze said.
“That is why B.C. will also be asking that the Provincial Renal Agency lead and work with other B.C. clinicians to evaluate the cumulative experience of B.C. and will be inviting other jurisdictions in Canada to participate.”
The Ministry of Health agrees that the cost of Soliris is too high. Heinze said that the manufacturer, Alexion, is charging Canadians 21 per cent more than other countries, like the UK. From 2010 to Q3 of 2017, the company reported revenues of $14.2 Billion U.S. from Soliris.
“In September 2017, the Patent Medicine Price Review Board found that the price Alexion Pharma set for its drug Soliris is excessive and ordered the company to pay back excessive revenues and lower its price to match the lowest of the other comparator countries,” Heinze said.
“B.C. supports the Patent Medicines Price Review Board’s findings. We understand that Alexion will be appealing the Review Boards decision and B.C. will be seeking to become actively involved in the appeal. We would also strongly encourage our partner jurisdictions and patient groups to lend their voice to this as well.
“The health of our public drug plans and our ability to help patients and families is dependent on us working together to hold companies like Alexion accountable for the outrageous prices they set. We will continue to take action to protect rare disease patients and public funders from exorbitant drug costs.”
ADVOCACY GROUPS OPTIMISTIC
The Better Pharmacare Coalition — an organization that represents 23 non-profit and charity health groups across the country, including aHUS Canada — is praising the B.C. government for their decision.
“I am very pleased that Minister Dix and the leadership of the BC PharmaCare Program have chosen to provide coverage for this life-saving medication in B.C.,” said Dr. Ganive Bhinder, executive director of the Better Pharmacare Coalition, in a press release issued Nov. 21.
“We know that there are enormous cost pressures in health, but the Minister has demonstrated an openness, and a willingness to listen to patients and we applaud the approach the Ministry has taken in this case.”
AHUS Canada is pleased as well, however they believe the process to get Soliris funded could have been much faster.
“We at aHUS Canada are very happy with the decision of the B.C. Ministry of Health to fund Soliris to aHUS patients on a case-by-case basis,” said Michael Eygenaam, vice-chair of aHUS Canada.
“We hope Shantee makes a full recovery — that her kidney function returns … She has been without Soliris far too long and we are concerned about permanent damage due to the delay.”
Eygenaam said their organization asked the previous Liberal government a few years ago to fund Soliris for aHUS patients, and had brought the issue froward to Dix as well in September.
“This delay was not necessary,” he said. “They could have had this ‘expensive drugs advisory committee’ in place before Shantee and others became ill in order for those patients to have had more timely access.”
Eygenaam believes that a program in B.C. should be created in collaboration with the health ministry, aHUS specialist physicians, patients receiving the treatment and their families, Alexion and aHUS Canada. In his opinion, England has the best system for access, using a country-wide expert centre with special diagnostic facilities staffed by aHUS experts.
“We want to be sure that the new committee is qualified, having extensive knowledge of aHUS and its treatment, making their decisions on the latest science. If they weigh their decisions mainly on cost, they will likely make unbalanced decisions, considering the cost too heavily over the specialist opinion,” Eygenaam said.
“We respect the province’s need to contain costs and will be monitoring that balance. We also want to be sure that any aHUS patient in B.C. who has lost their kidneys in the past due to aHUS have access to treatment that would allow them to have a successful kidney transplant. We will speak out for patients if they are denied coverage when they need it.”
miranda@langleytimes.com
Like us on Facebook and follow us on Twitter